Source:
https://www.sec.gov/Archives/edgar/data/1703647/000119312519239976/d72917ds1.htm
Reddit thread:
Frequency Therapeutics files IPO, first FX-322 results published! from tinnitus
Credit:
Thank you to Reddit user /u/pandoira for finding this update and bringing it to the attention of the tinnitus and hearing loss community!
Source:
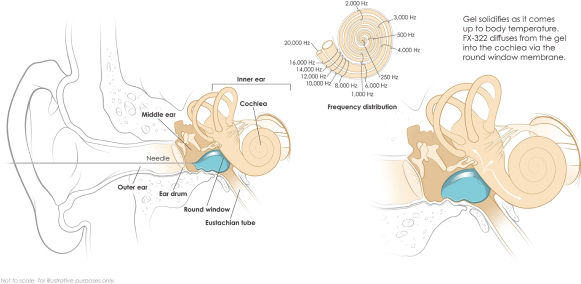
The image below shows delivery of FX-322 that turns to a gel in the middle ear. The drug diffuses into the cochlea and is expected to create the greatest concentration of drug in the high frequency region of the cochlea.
Clinical results
Phase 1/2 clinical trial
We conducted a Phase 1/2 clinical trial of FX-322 in which we enrolled 23 adult patients aged 33 to 64 with an established diagnosis of mild to moderately severe stable SNHL, defined as the average pure tone value of 26 to 70 dB at the 500, 1000, 2000 and 4000 Hz frequencies, who had no change of 10 db or more at any frequency for more than six months. Fourteen patients had mild SNHL and nine patients had moderate to moderately severe SNHL. Of the nine moderate to moderately severe patients, six were randomized to FX-322 and three to placebo. In this trial, 15 patients were treated with a single injection of FX-322 and eight patients received placebo. Each patient had a documented medical history consistent with either noise-induced hearing loss, or NIHL, typically from noise exposure at work, or sudden SNHL, or SSNHL, which is characterized as a loss of 30 dB at three adjacent frequencies occurring over a 72-hour period. All patients had stable SNHL, meaning their hearing function at study entry was not significantly different based on a documented audiogram from at least six months prior to the study. Hearing function, specifically speech intelligibility, was assessed using WR and WIN. Hearing loudness was also measured using pure tone audiometry. Patients were randomized to a single injection of FX-322 or placebo administered at one of two different dose volumes (0.05 mL and 0.2 mL) to assess the safety of FX-322 administration and systemic exposure to FX-322. Follow-up visits occurred at 15, 30, 60, and 90 days after injection.
The objectives of the trial were to assess:
| • | the systemic safety of FX-322; |
| • | the plasma pharmacokinetic profile to determine the systemic exposure to FX-322; and |
| • | the effect of FX-322 on measures of ear health and hearing function. |
FX-322 was observed to be well-tolerated in this trial. No serious adverse events were observed, and all treatment-related adverse events were mild, procedure-related, and generally resolved within minutes after dosing. We also observed limited concentrations of the FX-322 components in systemic circulation.
105
In addition to the prospective analysis, we conducted a prospective statistical analysis where we tested whether the Day 90 WR value for each patient fell outside of the 95% confidence interval compared to their baseline WR value. A confidence interval, or CI, is a range of values in which, statistically, there is a specified level of confidence where the result lies. In this patient-by-patient analysis, we observed statistically significant and clinically meaningful increases in WR in four of 15 patients treated with FX-322 at Day 90, who were among the six FX-322 patients that had moderate to moderately severe SNHL (shown in the figure below). There was no apparent association between WR improvements and whether the patients had stable NIHL, or stable SSNHL, and similar results were obtained with both dose volumes. This is consistent with published work showing drug delivery to the cochlea depends more on the concentration of the drug than the volume of injection. There were no clinically meaningful WR improvements observed in the placebo group.
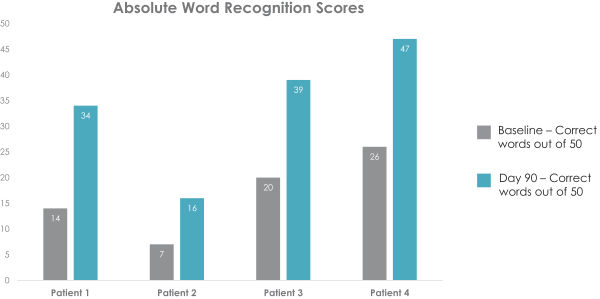
106
We also performed a post hoc analysis that showed a statistically significant improvement in WR by all FX-322-treated patients versus all placebo patients (p=0.01). A p value, as expressed in the data above, is the probability that the difference between two data sets was due to chance. The smaller the p value, the more likely the differences are not due to chance alone. In general, if the p value is less than or equal to 0.05, the outcome is statistically significant. The data are presented as adjusted mean relative percent change from baseline in the figure below. FX-322 treated patients saw improvements as early as 15 days after treatment that were sustained over 90 days.
Word Recognition
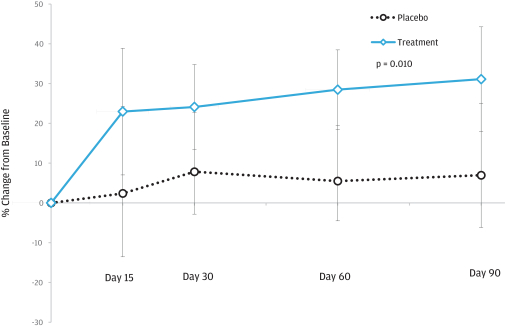
107
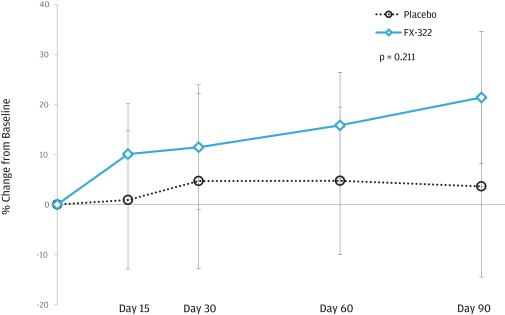
We performed an additional post hoc analysis on WIN data. As shown in the figure below, the adjusted mean relative percent change from baseline was assessed at 15, 30, 60, and 90 days after injection, and a trend in improvement was seen in FX-322-treated patients versus placebo. Also, there were non-statistically significant trends in improved WIN scores at Day 90 in the four FX-322, treated patients that had statistically significant and clinically meaningful improvements in WR in the prospective statistical analysis.
Words-In-Noise
We assessed audiometric changes from 250 Hz to 8000 Hz for all patients. Since drug enters closest to the high frequency region, the greatest drug exposure is expected to occur in the high frequency region. While no statistical differences were observed at any frequency when comparing pooled treatment groups, four of the moderate to moderately severe FX-322 patients showed a 10 dB threshold improvement at 8000 Hz at Day 90.
Planned Phase 2a clinical trial
Based on our analysis of the data from our Phase 1/2 clinical trial, we intend to initiate a randomized, double-blind, placebo-controlled, single- and repeat-dose Phase 2a clinical trial of FX-322 at approximately 12 sites in the United States in the fourth quarter of 2019. We plan to enroll approximately 96 adults aged 18 to 65 with stable SNHL. As in the Phase 1/2 clinical trial, patients must have a documented medical history consistent with either stable NIHL or stable SSNHL, with an average range of 26 to 70 dB loss measured by pure tone audiometry across four frequencies.
To explore how a single dose compares to multiple doses of FX-322, we plan to randomize patients to one of four groups, each of which will receive four injections, once per week at weekly intervals starting at the initial visit. Group 1 will receive one injection of FX-322 and three injections of placebo. Group two will receive two injections of FX-322 and two injections of placebo. Group three will receive four injections of FX-322. Group four will receive four injections of placebo. Patients will have follow-up visits two weeks after dosing and then monthly for seven months. The efficacy endpoints of this trial are expected to be WR, WIN, and pure tone audiometry in the range of 250 to 8000 Hz. The exploratory efficacy endpoints are expected to be the Tinnitus Functional Index, the Hearing Handicap Inventory for Adults, and pure tone audiometry in the range of 9000 to 16000 Hz. The selection of the efficacy endpoints in this study build on the learnings from the Phase 1/2 trial,
108
and we believe will add to our knowledge on the potential ways in which FX-322 may improve hearing function. We expect to report top-line data from this trial in the second half of 2020. We may also conduct clinical research in presbycusis.
Preclinical studies
Prior to commencing clinical trials, we tested FX-322 in multiple preclinical studies, including in human cells ex vivo and functional hearing tests in mice in vivo. In in vitro testing of isolated human inner ear progenitor cells with the compounds comprising FX-322, we observed the formation of new progenitor cells and their subsequent conversion into hair cells. We also observed translation across species in our in vitro studies of the inner ear progenitor cells from rhesus macaques in which a similar expansion of cell numbers were observed as in the in vitro studies of human cells.
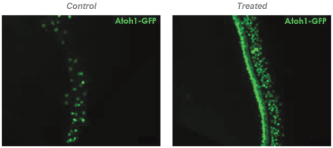
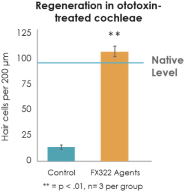
We also conducted ex vivo testing in intact cochlea isolated from mice. To cause hair cell loss, we exposed the cochlea for 16 hours to an aminoglycoside antibiotic that is toxic to hair cells. We then treated the cochlea for 72 hours with the compounds comprising the active agents in FX-322. Aminoglycoside treatment (left panel in the figure below) killed more than 80% of the hair cells in the cochlea (shown in green). By contrast, cochlea treated with the compounds in FX-322 (shown in the middle panel) regenerated hair cells to a near native level, as shown graphically in the right panel.
Restoration of Hair Cells in Mouse Cochlea
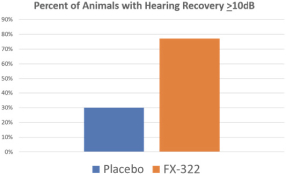
We also tested FX-322 in a mouse model of severe noise-induced hearing loss. Following noise exposure, 47 mice were treated with FX-322 and 37 were treated with placebo. Hearing function was measured using auditory brainstem response, or ABR, in which the signal generated by the auditory nerve upon sensing sound is detected by electrodes on the scalp. We performed ABR testing after 24 hours, and measured hearing recovery after 30 days. The figure below shows the percentage of mice treated with FX-322 (shown in orange) or with placebo (shown in blue) that achieved a hearing recovery of at least 10 dB at 20000 Hz, a mid-range
109
Table of Contents
frequency for mice. The improvement observed in the placebo-treated mice was due to recovery of temporary effects not related to hair cell death, which is typical following acute hearing loss.
Hearing Recovery in Mice Treated with FX-322
We have also conducted pharmacokinetic tests in multiple species in which we observed that FX-322 administration achieved therapeutic levels of the active pharmaceutical ingredients in the cochlea.